当前位置:
X-MOL 学术
›
J. Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Discovery of Novel Apigenin–Piperazine Hybrids as Potent and Selective Poly (ADP-Ribose) Polymerase-1 (PARP-1) Inhibitors for the Treatment of Cancer
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2021-08-18 , DOI: 10.1021/acs.jmedchem.1c00735
Huan Long 1 , Xiaolong Hu 1 , Baolin Wang 1 , Quan Wang 1 , Rong Wang 1 , Shumeng Liu 1 , Fei Xiong 2 , Zhenzhou Jiang 3 , Xiao-Qi Zhang 4 , Wen-Cai Ye 4 , Hao Wang 1
Journal of Medicinal Chemistry ( IF 6.8 ) Pub Date : 2021-08-18 , DOI: 10.1021/acs.jmedchem.1c00735
Huan Long 1 , Xiaolong Hu 1 , Baolin Wang 1 , Quan Wang 1 , Rong Wang 1 , Shumeng Liu 1 , Fei Xiong 2 , Zhenzhou Jiang 3 , Xiao-Qi Zhang 4 , Wen-Cai Ye 4 , Hao Wang 1
Affiliation
|
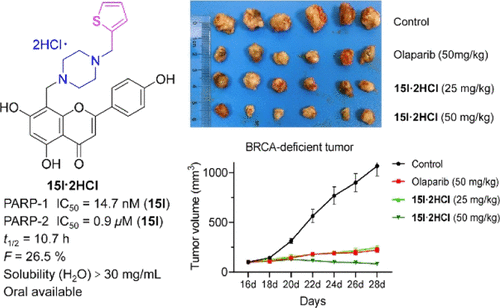
Poly (ADP-ribose) polymerase-1 (PARP-1) is a potential target for the discovery of chemosensitizers and anticancer drugs. Amentoflavone (AMF) is reported to be a selective PARP-1 inhibitor. Here, structural modifications and trimming of AMF have led to a series of AMF derivatives (9a–h) and apigenin–piperazine/piperidine hybrids (14a–p, 15a–p, 17a–h, and 19a–f), respectively. Among these compounds, 15l exhibited a potent PARP-1 inhibitory effect (IC50 = 14.7 nM) and possessed high selectivity to PARP-1 over PARP-2 (61.2-fold). Molecular dynamics simulation and the cellular thermal shift assay revealed that 15l directly bound to the PARP-1 structure. In in vitro and in vivo studies, 15l showed a potent chemotherapy sensitizing effect against A549 cells and a selective cytotoxic effect toward SK-OV-3 cells through PARP-1 inhibition. 15l·2HCl also displayed good ADME characteristics, pharmacokinetic parameters, and a desirable safety margin. These findings demonstrated that 15l·2HCl may serve as a lead compound for chemosensitizers and the (BRCA-1)-deficient cancer therapy.
中文翻译:

发现新型芹菜素-哌嗪杂合体作为有效的选择性聚(ADP-核糖)聚合酶-1(PARP-1)抑制剂用于治疗癌症
聚(ADP-核糖)聚合酶-1 (PARP-1) 是发现化学增敏剂和抗癌药物的潜在靶标。据报道,Amentoflavone ( AMF ) 是一种选择性 PARP-1 抑制剂。在这里, AMF的结构修饰和修剪分别产生了一系列AMF衍生物 ( 9a–h ) 和芹菜素-哌嗪/哌啶杂化物 ( 14a–p 、 15a–p 、 17a–h和19a–f )。在这些化合物中, 15l表现出有效的PARP-1抑制作用(IC 50 = 14.7 nM),并且对PARP-1的选择性高于对PARP-2的选择性(61.2倍)。分子动力学模拟和细胞热位移测定表明15l直接与PARP-1结构结合。在体外和体内研究中, 15l对 A549 细胞显示出有效的化疗增敏作用,并通过 PARP-1 抑制对 SK-OV-3 细胞具有选择性细胞毒作用。 15l·2HCl还表现出良好的 ADME 特性、药代动力学参数和理想的安全裕度。这些发现表明15l·2HCl可作为化学增敏剂和 (BRCA-1) 缺陷型癌症治疗的先导化合物。
更新日期:2021-08-26
中文翻译:

发现新型芹菜素-哌嗪杂合体作为有效的选择性聚(ADP-核糖)聚合酶-1(PARP-1)抑制剂用于治疗癌症
聚(ADP-核糖)聚合酶-1 (PARP-1) 是发现化学增敏剂和抗癌药物的潜在靶标。据报道,Amentoflavone ( AMF ) 是一种选择性 PARP-1 抑制剂。在这里, AMF的结构修饰和修剪分别产生了一系列AMF衍生物 ( 9a–h ) 和芹菜素-哌嗪/哌啶杂化物 ( 14a–p 、 15a–p 、 17a–h和19a–f )。在这些化合物中, 15l表现出有效的PARP-1抑制作用(IC 50 = 14.7 nM),并且对PARP-1的选择性高于对PARP-2的选择性(61.2倍)。分子动力学模拟和细胞热位移测定表明15l直接与PARP-1结构结合。在体外和体内研究中, 15l对 A549 细胞显示出有效的化疗增敏作用,并通过 PARP-1 抑制对 SK-OV-3 细胞具有选择性细胞毒作用。 15l·2HCl还表现出良好的 ADME 特性、药代动力学参数和理想的安全裕度。这些发现表明15l·2HCl可作为化学增敏剂和 (BRCA-1) 缺陷型癌症治疗的先导化合物。































 京公网安备 11010802027423号
京公网安备 11010802027423号