当前位置:
X-MOL 学术
›
Chem. Eur. J.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Cascade Reaction to Selectively Synthesize Multifunctional Indole Derivatives by IrIII-Catalyzed C−H Activation
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2021-08-08 , DOI: 10.1002/chem.202101602 Xin-Yue Chai 1 , Hui-Bei Xu 1 , Lin Dong 1
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2021-08-08 , DOI: 10.1002/chem.202101602 Xin-Yue Chai 1 , Hui-Bei Xu 1 , Lin Dong 1
Affiliation
|
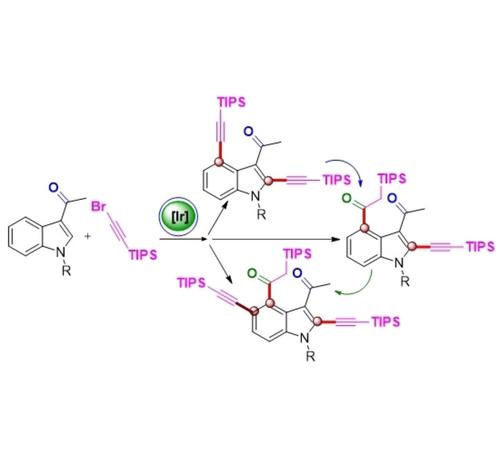
An effective and condition-controlled way to synthesize with high selectivity a variety of functionalized indoles with potent biological properties has been developed. Notably, 2,4-dialkynyl indole products were obtained by direct double C−H bond alkynylation, whereas alkynyl at the C4 position could convert to carbonyl to generate 2-alkynyl-3,4-diacetyl indoles fast and effectively. Additionally, a one-pot relay catalytic reaction led to 2,5-di-alkynyl-3,4-diacetyl indoles when using a carbonyl group as the directing group and by controlling the type and quantity of additives. A possible mechanism was proposed based on many studies including deuterium-exchange experiments, the necessary conditions of product conversion, and the effect of water on the reaction.
中文翻译:

通过 IrIII 催化的 C-H 活化选择性合成多功能吲哚衍生物的级联反应
已经开发出一种有效且条件控制的方法来以高选择性合成各种具有有效生物特性的功能化吲哚。值得注意的是,2,4-二炔基吲哚产物是通过直接双C-H键炔基化获得的,而C4位的炔基可以快速有效地转化为羰基生成2-炔基-3,4-二乙酰基吲哚。此外,当使用羰基作为导向基团并通过控制添加剂的类型和数量时,一锅接力催化反应产生了 2,5-二炔基-3,4-二乙酰吲哚。基于许多研究,包括氘交换实验、产物转化的必要条件以及水对反应的影响,提出了一种可能的机制。
更新日期:2021-09-15
中文翻译:

通过 IrIII 催化的 C-H 活化选择性合成多功能吲哚衍生物的级联反应
已经开发出一种有效且条件控制的方法来以高选择性合成各种具有有效生物特性的功能化吲哚。值得注意的是,2,4-二炔基吲哚产物是通过直接双C-H键炔基化获得的,而C4位的炔基可以快速有效地转化为羰基生成2-炔基-3,4-二乙酰基吲哚。此外,当使用羰基作为导向基团并通过控制添加剂的类型和数量时,一锅接力催化反应产生了 2,5-二炔基-3,4-二乙酰吲哚。基于许多研究,包括氘交换实验、产物转化的必要条件以及水对反应的影响,提出了一种可能的机制。















































 京公网安备 11010802027423号
京公网安备 11010802027423号