当前位置:
X-MOL 学术
›
Acc. Chem. Res.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Heterogeneous Molecular Catalysts of Metal Phthalocyanines for Electrochemical CO2 Reduction Reactions
Accounts of Chemical Research ( IF 16.4 ) Pub Date : 2021-08-04 , DOI: 10.1021/acs.accounts.1c00200 Yueshen Wu 1, 2 , Yongye Liang 3, 4 , Hailiang Wang 1, 2
Accounts of Chemical Research ( IF 16.4 ) Pub Date : 2021-08-04 , DOI: 10.1021/acs.accounts.1c00200 Yueshen Wu 1, 2 , Yongye Liang 3, 4 , Hailiang Wang 1, 2
Affiliation
|
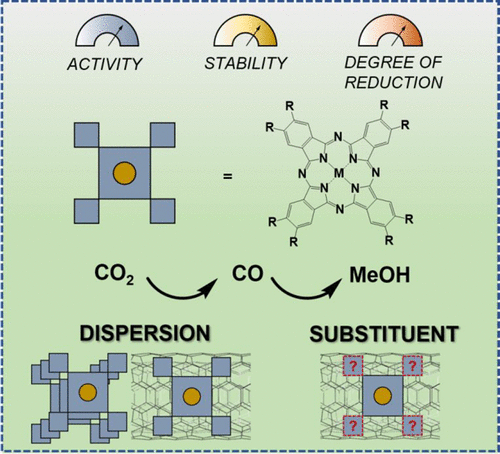
Molecular catalysts, often deployed in homogeneous conditions, are favorable systems for structure–reactivity correlation studies of electrochemical reactions because of their well-defined active site structures and ease of mechanistic investigation. In pursuit of selective and active electrocatalysts for the CO2 reduction reactions which are promising for converting carbon emissions to useful fuels and chemical products, it is desirable to support molecular catalysts on substrates because heterogeneous catalysts can afford the high current density and operational convenience that practical electrolyzers require. Herein, we share our understanding in the development of heterogenized metal phthalocyanine catalysts for the electrochemical reduction of CO2. From the optimization of preparation methods and material structures for the electrocatalytic activity toward CO2 reduction to CO, we find that molecular-level dispersion of the active material and high electrical conductivity of the support are among the most important factors controlling the activity. The molecular nature of the active site enables mechanism-based optimization. We demonstrate how electron-withdrawing and -donating ligand substituents can be utilized to modify the redox property of the molecule and improve its catalytic activity and stability. Adjusting these factors further allows us to achieve electrochemical reduction of CO2 to methanol with appreciable activity, which has not been attainable by conventional molecular catalysts. The six-electron reduction process goes through CO as the key intermediate. Rapid and continuous electron delivery to the active site favors further reduction of CO to methanol. We also point out that, in homogeneous electrocatalysis where the catalyst molecules are dissolved in the electrolyte solution, even if the molecular structure remains intact, the actual catalysis may be dominated by molecules permanently adsorbed on the electrode surface and is thus heterogeneous in nature. This account uses our research on CO2 electroreduction reactions catalyzed by metal phthalocyanine molecules to illustrate our understanding about heterogeneous molecular electrocatalysis, which is also applicable to other electrochemical systems.
中文翻译:

用于电化学 CO2 还原反应的金属酞菁多相分子催化剂
分子催化剂通常在均相条件下使用,由于其明确的活性位点结构和易于机理研究,是电化学反应结构-反应性相关研究的有利系统。为了寻求用于CO 2还原反应的选择性和活性电催化剂,其有望将碳排放转化为有用的燃料和化学产品,需要在基底上负载分子催化剂,因为非均相催化剂可以提供实际所需的高电流密度和操作便利性。电解槽需要。在此,我们分享了我们对开发用于电化学还原CO 2的多相金属酞菁催化剂的理解。通过对CO 2还原为CO的电催化活性的制备方法和材料结构的优化,我们发现活性材料的分子水平分散和载体的高电导率是控制活性的最重要因素之一。活性位点的分子性质使得基于机制的优化成为可能。我们展示了如何利用吸电子和供电子配体取代基来改变分子的氧化还原性质并提高其催化活性和稳定性。进一步调整这些因素使我们能够以可观的活性实现CO 2电化学还原为甲醇,这是传统分子催化剂无法实现的。六电子还原过程以CO为关键中间体。快速、连续的电子传递到活性位点有利于 CO 进一步还原为甲醇。 我们还指出,在催化剂分子溶解在电解质溶液中的均相电催化中,即使分子结构保持完整,实际的催化作用也可能由永久吸附在电极表面上的分子主导,因此本质上是非均相的。本文通过我们对金属酞菁分子催化的CO 2电还原反应的研究来说明我们对非均相分子电催化的理解,这也适用于其他电化学系统。
更新日期:2021-08-17
中文翻译:

用于电化学 CO2 还原反应的金属酞菁多相分子催化剂
分子催化剂通常在均相条件下使用,由于其明确的活性位点结构和易于机理研究,是电化学反应结构-反应性相关研究的有利系统。为了寻求用于CO 2还原反应的选择性和活性电催化剂,其有望将碳排放转化为有用的燃料和化学产品,需要在基底上负载分子催化剂,因为非均相催化剂可以提供实际所需的高电流密度和操作便利性。电解槽需要。在此,我们分享了我们对开发用于电化学还原CO 2的多相金属酞菁催化剂的理解。通过对CO 2还原为CO的电催化活性的制备方法和材料结构的优化,我们发现活性材料的分子水平分散和载体的高电导率是控制活性的最重要因素之一。活性位点的分子性质使得基于机制的优化成为可能。我们展示了如何利用吸电子和供电子配体取代基来改变分子的氧化还原性质并提高其催化活性和稳定性。进一步调整这些因素使我们能够以可观的活性实现CO 2电化学还原为甲醇,这是传统分子催化剂无法实现的。六电子还原过程以CO为关键中间体。快速、连续的电子传递到活性位点有利于 CO 进一步还原为甲醇。 我们还指出,在催化剂分子溶解在电解质溶液中的均相电催化中,即使分子结构保持完整,实际的催化作用也可能由永久吸附在电极表面上的分子主导,因此本质上是非均相的。本文通过我们对金属酞菁分子催化的CO 2电还原反应的研究来说明我们对非均相分子电催化的理解,这也适用于其他电化学系统。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号