当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Rhodium-Catalyzed Denitrogenative Transannulation of N-Sulfonyl-1,2,3-triazoles with Glycals Giving Pyrroline-Fused N-Glycosides
Organic Letters ( IF 4.9 ) Pub Date : 2021-08-04 , DOI: 10.1021/acs.orglett.1c02141 Jingjing Bi 1 , Qiang Tan 1 , Hao Wu 1 , Qingfeng Liu 1 , Guisheng Zhang 1
Organic Letters ( IF 4.9 ) Pub Date : 2021-08-04 , DOI: 10.1021/acs.orglett.1c02141 Jingjing Bi 1 , Qiang Tan 1 , Hao Wu 1 , Qingfeng Liu 1 , Guisheng Zhang 1
Affiliation
|
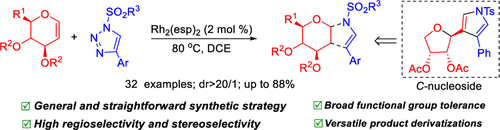
Described here is a selective synthesis of 2,3-dihydropyrrole-fused N-glycosides through rhodium-catalyzed denitrogenative transannulation of N-sulfonyl-1,2,3-triazoles with glycals. A series of pyrroline-fused N-glycosides are afforded in moderate to excellent yields with exclusive regioselectivity and stereoselectivity. Functional application of such a resultant product by oxidative addition and epoxidation is also explored. Notably, the treatment of a pyrroline-fused N-glycoside (3a) with TMSOTf efficiently leads to an interesting unexpected C-nucleoside (9) via a TMSOTf-inducing ring opening/acetyl migration/ring closing reaction sequence.
中文翻译:

N-磺酰基-1,2,3-三唑的铑催化脱氮环化反应,得到吡咯啉融合的 N-糖苷
这里描述的是通过铑催化的N-磺酰基-1,2,3-三唑与聚糖的脱氮环化反应选择性合成 2,3-二氢吡咯稠合的N-糖苷。一系列吡咯啉稠合的N-糖苷以中等至极好的收率提供,具有独特的区域选择性和立体选择性。还探索了这种通过氧化加成和环氧化得到的产物的功能应用。值得注意的是,通过 TMSOTf 诱导的开环/乙酰基迁移/闭环反应序列,用 TMSOTf处理吡咯啉稠合的N-糖苷(3a)有效地导致有趣的意外C-核苷(9)。
更新日期:2021-08-20
中文翻译:

N-磺酰基-1,2,3-三唑的铑催化脱氮环化反应,得到吡咯啉融合的 N-糖苷
这里描述的是通过铑催化的N-磺酰基-1,2,3-三唑与聚糖的脱氮环化反应选择性合成 2,3-二氢吡咯稠合的N-糖苷。一系列吡咯啉稠合的N-糖苷以中等至极好的收率提供,具有独特的区域选择性和立体选择性。还探索了这种通过氧化加成和环氧化得到的产物的功能应用。值得注意的是,通过 TMSOTf 诱导的开环/乙酰基迁移/闭环反应序列,用 TMSOTf处理吡咯啉稠合的N-糖苷(3a)有效地导致有趣的意外C-核苷(9)。

















































 京公网安备 11010802027423号
京公网安备 11010802027423号