Tetrahedron ( IF 2.1 ) Pub Date : 2021-07-16 , DOI: 10.1016/j.tet.2021.132353 Yoshino Fujii 1 , Yuta Taguchi 1 , Shugo Tokai 1 , Yasuhiro Matsumoto 1 , Naruhiro Yoshida 1 , Tetsuo Iwasawa 1
|
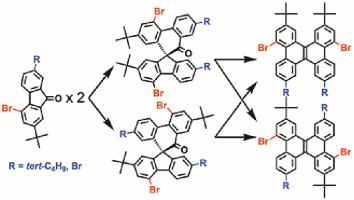
In this paper we explored several issues surrounding metal-free selective synthesis of dibenzo[g,p]chrysene (DBC) by steps that consist of the dimerization of fluorenones and the carbocation 1,2-shift. We focus on structurally non-symmetric two fluorenones having a bromine atom as a reactive moiety and a tert-butyl group as a solubilizing agent: One is 4-bromo-2,7-di-tert-butyl-9-fluorenone (1), and the other 4,7-dibromo-2-(tert-butyl)-9-fluorenone (2). Each starting 1 and 2 undertakes the homo-coupling to give two isomeric spiroketones and the following Wagner-Meerwein rearrangement to yield two isomeric DBCs. The spiroketone intermediates have now been crystallographically characterized, which informed us of the exact structures and lead us to comprehend which aryl group predominantly migrates from one carbon to a neighboring carbon along with production of DBC skeletons. Although the construction of DBCs proved to be involved in four paths, it seems likely now that a clear demonstration of making different DBCs by different reaction conditions enables us to accomplish selective synthesis of DBCs.
中文翻译:

二苯并[g,p]芘衍生物选择性合成产率的相关分析
在本文中,我们通过芴酮二聚和碳正离子 1,2-转变的步骤探讨了围绕无金属选择性合成二苯并 [ g , p ]chrysene (DBC) 的几个问题。我们专注于结构不对称的两种芴酮,它们具有一个溴原子作为反应部分和一个叔丁基作为增溶剂:一个是 4-bromo-2,7-di- tert -butyl-9-芴酮 ( 1 ) ,以及其他 4,7-dibromo-2-( tert -butyl)-9-芴酮 ( 2 )。每个开始1和2进行同源偶联以产生两个异构体螺酮和以下 Wagner-Meerwein 重排以产生两个异构体 DBC。现在已经对螺酮中间体进行了晶体学表征,这使我们了解了确切的结构,并引导我们理解哪个芳基主要从一个碳迁移到相邻的碳以及 DBC 骨架的产生。尽管已证明 DBC 的构建涉及四个途径,但现在看来,通过不同反应条件制备不同 DBC 的明确证明使我们能够完成 DBC 的选择性合成。






































 京公网安备 11010802027423号
京公网安备 11010802027423号