当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Twofold Radical-Based Synthesis of N,C-Difunctionalized Bicyclo[1.1.1]pentanes
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2021-06-23 , DOI: 10.1021/jacs.1c04180 Helena D Pickford 1 , Jeremy Nugent 1 , Benjamin Owen 1 , James J Mousseau 2 , Russell C Smith 3 , Edward A Anderson 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2021-06-23 , DOI: 10.1021/jacs.1c04180 Helena D Pickford 1 , Jeremy Nugent 1 , Benjamin Owen 1 , James J Mousseau 2 , Russell C Smith 3 , Edward A Anderson 1
Affiliation
|
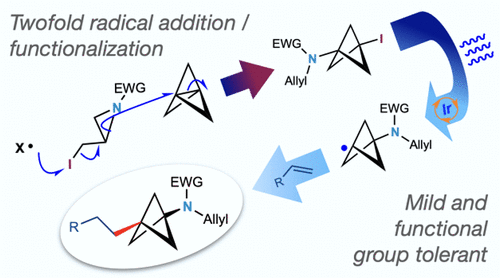
Bicyclo[1.1.1]pentylamines (BCPAs) are of growing importance to the pharmaceutical industry as sp3-rich bioisosteres of anilines and N-tert-butyl groups. Here we report a facile synthesis of 1,3-disubstituted BCPAs using a twofold radical functionalization strategy. Sulfonamidyl radicals, generated through fragmentation of α-iodoaziridines, undergo initial addition to [1.1.1]propellane to afford iodo-BCPAs; the newly formed C–I bond in these products is then functionalized via a silyl-mediated Giese reaction. This chemistry also translates smoothly to 1,3-disubstituted iodo-BCPs. A wide variety of radical acceptors and iodo-BCPAs are accommodated, providing straightforward access to an array of valuable aniline-like isosteres.
中文翻译:

N,C-双官能化双环[1.1.1]戊烷的双重自由基合成
双环[1.1.1] pentylamines(BCPAs)是重要性日益增加,以制药业作为藻3个苯胺和富含的bioisosters ñ -叔丁基的基团。在这里,我们报告了使用双重自由基功能化策略轻松合成 1,3-二取代 BCPA。磺胺基自由基,通过 α-碘氮丙啶的碎裂产生,经过初始添加到 [1.1.1] 丙烷中,得到碘-BCPA;然后通过甲硅烷基介导的 Giese 反应将这些产物中新形成的 C-I 键功能化。这种化学反应也可以顺利转化为 1,3-二取代碘-BCP。可容纳多种自由基受体和碘-BCPA,可直接获取一系列有价值的类苯胺等排体。
更新日期:2021-07-07
中文翻译:

N,C-双官能化双环[1.1.1]戊烷的双重自由基合成
双环[1.1.1] pentylamines(BCPAs)是重要性日益增加,以制药业作为藻3个苯胺和富含的bioisosters ñ -叔丁基的基团。在这里,我们报告了使用双重自由基功能化策略轻松合成 1,3-二取代 BCPA。磺胺基自由基,通过 α-碘氮丙啶的碎裂产生,经过初始添加到 [1.1.1] 丙烷中,得到碘-BCPA;然后通过甲硅烷基介导的 Giese 反应将这些产物中新形成的 C-I 键功能化。这种化学反应也可以顺利转化为 1,3-二取代碘-BCP。可容纳多种自由基受体和碘-BCPA,可直接获取一系列有价值的类苯胺等排体。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号