当前位置:
X-MOL 学术
›
Chem. Soc. Rev.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Recent advances in the chemistry of ketyl radicals
Chemical Society Reviews ( IF 40.4 ) Pub Date : 2021-3-23 , DOI: 10.1039/d0cs00358a
Áron Péter 1 , Soumitra Agasti , Oliver Knowles , Emma Pye , David J Procter
Chemical Society Reviews ( IF 40.4 ) Pub Date : 2021-3-23 , DOI: 10.1039/d0cs00358a
Áron Péter 1 , Soumitra Agasti , Oliver Knowles , Emma Pye , David J Procter
Affiliation
|
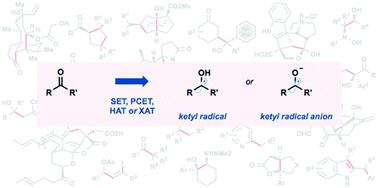
Ketyl radicals are valuable reactive intermediates for synthesis and are used extensively to construct complex, functionalized products from carbonyl substrates. Single electron transfer (SET) reduction of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of aldehydes and ketones is the classical approach for the formation of ketyl radicals and metal reductants are the archetypal reagents employed. The past decade has, however, witnessed significant advances in the generation and harnessing of ketyl radicals. This tutorial review highlights recent, exciting developments in the chemistry of ketyl radicals by comparing the varied contemporary – for example, using photoredox catalysts – and more classical approaches for the generation and use of ketyl radicals. The review will focus on different strategies for ketyl radical generation, their creative use in new synthetic protocols, strategies for the control of enantioselectivity, and detailed mechanisms where appropriate.
O bond of aldehydes and ketones is the classical approach for the formation of ketyl radicals and metal reductants are the archetypal reagents employed. The past decade has, however, witnessed significant advances in the generation and harnessing of ketyl radicals. This tutorial review highlights recent, exciting developments in the chemistry of ketyl radicals by comparing the varied contemporary – for example, using photoredox catalysts – and more classical approaches for the generation and use of ketyl radicals. The review will focus on different strategies for ketyl radical generation, their creative use in new synthetic protocols, strategies for the control of enantioselectivity, and detailed mechanisms where appropriate.
中文翻译:

酮基自由基化学的最新进展
酮基自由基是有价值的合成反应中间体,广泛用于从羰基底物构建复杂的功能化产物。C的单电子转移(SET)还原![[双键,长度为 m-dash]](https://www.rsc.org/images/entities/char_e001.gif) 醛和酮的 O 键是形成酮基自由基的经典方法,金属还原剂是使用的原型试剂。然而,过去十年见证了酮基自由基的产生和利用方面的重大进展。本教程回顾通过比较各种当代(例如,使用光氧化还原催化剂)和更经典的生成和使用酮基自由基的方法,重点介绍了酮基自由基化学中最近令人兴奋的发展。审查将重点关注不同的酮基自由基生成策略、它们在新合成方案中的创造性应用、控制对映选择性的策略以及适当的详细机制。
醛和酮的 O 键是形成酮基自由基的经典方法,金属还原剂是使用的原型试剂。然而,过去十年见证了酮基自由基的产生和利用方面的重大进展。本教程回顾通过比较各种当代(例如,使用光氧化还原催化剂)和更经典的生成和使用酮基自由基的方法,重点介绍了酮基自由基化学中最近令人兴奋的发展。审查将重点关注不同的酮基自由基生成策略、它们在新合成方案中的创造性应用、控制对映选择性的策略以及适当的详细机制。
更新日期:2021-03-23
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond of aldehydes and ketones is the classical approach for the formation of ketyl radicals and metal reductants are the archetypal reagents employed. The past decade has, however, witnessed significant advances in the generation and harnessing of ketyl radicals. This tutorial review highlights recent, exciting developments in the chemistry of ketyl radicals by comparing the varied contemporary – for example, using photoredox catalysts – and more classical approaches for the generation and use of ketyl radicals. The review will focus on different strategies for ketyl radical generation, their creative use in new synthetic protocols, strategies for the control of enantioselectivity, and detailed mechanisms where appropriate.
O bond of aldehydes and ketones is the classical approach for the formation of ketyl radicals and metal reductants are the archetypal reagents employed. The past decade has, however, witnessed significant advances in the generation and harnessing of ketyl radicals. This tutorial review highlights recent, exciting developments in the chemistry of ketyl radicals by comparing the varied contemporary – for example, using photoredox catalysts – and more classical approaches for the generation and use of ketyl radicals. The review will focus on different strategies for ketyl radical generation, their creative use in new synthetic protocols, strategies for the control of enantioselectivity, and detailed mechanisms where appropriate.
中文翻译:

酮基自由基化学的最新进展
酮基自由基是有价值的合成反应中间体,广泛用于从羰基底物构建复杂的功能化产物。C的单电子转移(SET)还原
![[双键,长度为 m-dash]](https://www.rsc.org/images/entities/char_e001.gif) 醛和酮的 O 键是形成酮基自由基的经典方法,金属还原剂是使用的原型试剂。然而,过去十年见证了酮基自由基的产生和利用方面的重大进展。本教程回顾通过比较各种当代(例如,使用光氧化还原催化剂)和更经典的生成和使用酮基自由基的方法,重点介绍了酮基自由基化学中最近令人兴奋的发展。审查将重点关注不同的酮基自由基生成策略、它们在新合成方案中的创造性应用、控制对映选择性的策略以及适当的详细机制。
醛和酮的 O 键是形成酮基自由基的经典方法,金属还原剂是使用的原型试剂。然而,过去十年见证了酮基自由基的产生和利用方面的重大进展。本教程回顾通过比较各种当代(例如,使用光氧化还原催化剂)和更经典的生成和使用酮基自由基的方法,重点介绍了酮基自由基化学中最近令人兴奋的发展。审查将重点关注不同的酮基自由基生成策略、它们在新合成方案中的创造性应用、控制对映选择性的策略以及适当的详细机制。


































 京公网安备 11010802027423号
京公网安备 11010802027423号