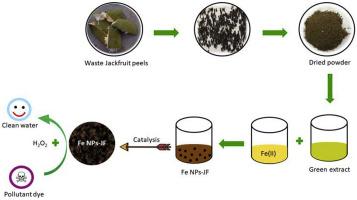
利用某些绿色化学原理合成纳米颗粒为纳米技术提供了一种可行且可持续的方法。用面包果合成铁纳米颗粒(Fe NPs)(波罗蜜)果皮提取物。具有高抗氧化剂含量的果皮可作为有价值生物分子的潜在来源,这些生物分子可作为绿色还原纳米粒子的生物还原剂,加帽剂和稳定剂。该方法除了使用无毒反应物材料并具有成本效益之外,还有效地利用了废物,并且在该过程中减少了废物的积累。利用傅里叶变换红外光谱(FTIR),透射电子显微镜(TEM),X射线衍射(XRD),配备能量色散的扫描电子显微镜(SEM)对使用菠萝蜜提取物(Fe NPs-JF)合成的铁纳米颗粒进行了表征。 X射线(EDX)技术。如此合成的纳米粒子由零价铁纳米粒子(nZVI)以及氧化铁和羟基氧化铁组成,平均大小为33 nm。进一步,合成的纳米颗粒作为类Fenton催化剂的功效已在诸如Fuchsin Basic之类的污染性染料的降解中得到证明。实验表明,这些铁纳米颗粒在318 K的初始20分钟内表现出优异的催化活性,去除效率高达87.5%。此外,去除品红碱性染料非常适合拟一阶模型。基于使用Arrhenius方程式计算的染料的氧化降解的活化能,可以推断出染料降解遵循表面控制的反应,其中限速步骤构成了表面化学反应而不是扩散。实验表明,这些铁纳米颗粒在318 K的初始20分钟内表现出优异的催化活性,去除效率高达87.5%。此外,去除品红碱性染料非常适合拟一阶模型。基于使用Arrhenius方程式计算的染料的氧化降解的活化能,可以推断出染料降解遵循表面控制的反应,其中限速步骤构成了表面化学反应而不是扩散。实验表明,这些铁纳米颗粒在318 K的初始20分钟内表现出优异的催化活性,去除效率高达87.5%。此外,去除品红碱性染料非常适合拟一阶模型。基于使用Arrhenius方程式计算的染料的氧化降解的活化能,可以推断出染料降解遵循表面控制的反应,其中限速步骤构成了表面化学反应而不是扩散。
 "点击查看英文标题和摘要"
"点击查看英文标题和摘要"
Green synthesis of iron nanoparticles using Artocarpus heterophyllus peel extract and their application as a heterogeneous Fenton-like catalyst for the degradation of Fuchsin Basic dye
Synthesis of nanoparticles by utilizing some of the green chemistry principles offers a viable and sustainable approach for nanotechnology. Iron nanoparticles (Fe NPs) were synthesised using Artocarpus heterophyllus (Jackfruit) peel extract. The peel, with its high antioxidant content, serves as a potential source of valuable biomolecules which act as the bio-reductants, capping and stabilizing agents for green synthesis of nanoparticles. The method, apart from using non toxic reactant materials and being cost effective, utilizes waste usefully and in the process reduces waste accumulation. The iron nanoparticles synthesised using jackfruit peel extract (Fe NPs-JF) were characterized by Fourier transform infrared spectroscopy (FTIR), Transmission electron microscopy (TEM), X-Ray diffraction (XRD), Scanning electron microscopy (SEM) equipped with energy dispersive X-ray (EDX) techniques. The nanoparticles so synthesised comprised of zero-valent iron nanoparticles (nZVI) along with iron oxides and oxyhydroxide, with an average size of 33 nm. Further, the efficacy of synthesised nanoparticles as Fenton-like catalyst has been demonstrated in the degradation of pollutant dyes like Fuchsin Basic. The experiment showed that these iron nanoparticles exhibited excellent catalytic activity with a high removal efficiency of 87.5% in initial 20 min at 318 K. In addition, removal of Fuchsin Basic dye fitted well to be a pseudo-first order model. On the basis of the activation energy for the oxidative degradation of the dye calculated using Arrhenius equation, it could be inferred that dye degradation followed a surface-controlled reaction wherein the rate-limiting step constituted a surface-chemical reaction instead of diffusion.














































 京公网安备 11010802027423号
京公网安备 11010802027423号