Bioorganic & Medicinal Chemistry Letters ( IF 2.5 ) Pub Date : 2020-11-17 , DOI: 10.1016/j.bmcl.2020.127687 Jana Waltemate 1 , Igor Ivanov 2 , Jahan B Ghasemi 3 , Elham Aghaee 3 , Constantin Gabriel Daniliuc 4 , Klaus Müller 1 , Helge Prinz 1
|
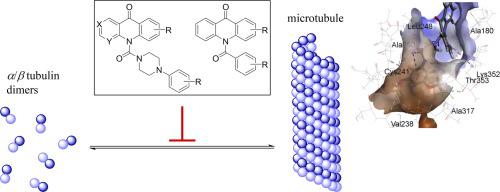
As part of our continuing search for potent inhibitors of tubulin polymerization, two novel series of 42 10-(4-phenylpiperazine-1-carbonyl)acridin-9(10H)-ones and N-benzoylated acridones were synthesized on the basis of a retrosynthetic approach. All newly synthesized compounds were tested for antiproliferative activity and interaction with tubulin. Several analogs potently inhibited tumor cell growth. Among the compounds tested, 10-(4-(3-methoxyphenyl)piperazine-1-carbonyl)acridin-9(10H)-one (17c) exhibited excellent growth inhibitory effects on 93 tumor cell lines, with an average GI50 value of 5.4 nM. We were able to show that the strong cytotoxic effects are caused by disruption of tubulin polymerization, as supported by the EBI (N,N'-Ethylenebis(iodoacetamide)) assay and the fact that the most potent inhibitors of cancer cell growth turned out to be the most efficacious tubulin polymerization inhibitors. Potencies were nearly comparable or superior to those of the antimitotic reference compounds. Closely related to this, the most active analogs inhibited cell cycling at the G2/M phase at concentrations down to 30 nM and induced apoptosis in K562 leukemia cells. We believe that our work not only proves the excellent suitability of the acridone scaffold for the design of potent tubulin polymerization inhibitors but also enables synthetic access to further potentially interesting N-acylated acridones.
中文翻译:

10-(4-Phenylpiperazine-1-carbonyl)acridin-9(10H)-ones 和相关化合物:微管蛋白聚合的合成、抗增殖活性和抑制
作为我们继续寻找微管蛋白聚合的有效抑制剂的一部分,基于 42 10-(4-苯基哌嗪-1-羰基)吖啶-9(10 H )-酮和N-苯甲酰化吖啶酮的两个新系列被合成逆合成方法。测试所有新合成的化合物的抗增殖活性和与微管蛋白的相互作用。几种类似物有效抑制肿瘤细胞生长。在测试的化合物中,10-(4-(3-甲氧基苯基)哌嗪-1-羰基)吖啶-9(10 H)-one ( 17c ) 对 93 种肿瘤细胞系表现出优异的生长抑制作用,平均 GI 50值 5.4 nM。我们能够证明强烈的细胞毒性作用是由微管蛋白聚合的破坏引起的,这得到了 EBI ( N , N'-亚乙基双(碘乙酰胺))测定以及癌细胞生长最有效的抑制剂被证明是最有效的微管蛋白聚合抑制剂这一事实。效力几乎可比或优于抗有丝分裂参考化合物的效力。与此密切相关的是,最活跃的类似物以低至 30 nM 的浓度抑制 G2/M 期的细胞循环并诱导 K562 白血病细胞凋亡。我们相信,我们的工作不仅证明了吖啶酮支架在设计有效的微管蛋白聚合抑制剂方面的出色适用性,而且还能够合成获得更多潜在有趣的N-酰化吖啶酮。





















































 京公网安备 11010802027423号
京公网安备 11010802027423号