当前位置:
X-MOL 学术
›
Appl. Surf. Sci.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
A recyclable molten-salt synthesis of B and K co-doped g-C3N4 for photocatalysis of overall water vapor splitting
Applied Surface Science ( IF 6.3 ) Pub Date : 2021-01-01 , DOI: 10.1016/j.apsusc.2020.148014 Ji-Chao Wang , Yuxia Hou , Fu-Dong Feng , Wen-Xia Wang , Weina Shi , Wanqing Zhang , Yu Li , Huihui Lou , Cheng-Xing Cui
Applied Surface Science ( IF 6.3 ) Pub Date : 2021-01-01 , DOI: 10.1016/j.apsusc.2020.148014 Ji-Chao Wang , Yuxia Hou , Fu-Dong Feng , Wen-Xia Wang , Weina Shi , Wanqing Zhang , Yu Li , Huihui Lou , Cheng-Xing Cui
|
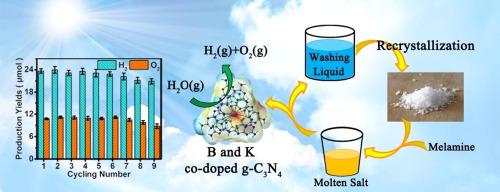
Abstract Photocatalytic overall water splitting is one of the green and efficient energy technologies. Due to difficult release of O2 from photocatalysts, the simultaneous generation of H2 and O2 is a critical challenge for water splitting. B and K co-doped g-C3N4 (B/K-g-C3N4) catalyst was synthesized by the recyclable molten-salt method in this study. Compared with the K doped and pristine g-C3N4 samples, B/K-g-C3N4 exhibited the highest photocatalytic activity for water vapor splitting under visible light illumination. The yield ratio of H2 and O2 reached approximately 2:1, and the corresponding rates were 1.18 and 0.58 μmol/h after 24 h of illumination. The B/K-g-C3N4 sample demonstrated good photocatalytic stability after 9 cycles. Additionally, the co-doped g-C3N4 catalyst, obtained from the repeatedly recyclable salts, still exhibited equivalent photocatalytic activity. Based on the built structure model with g-C3N4, the HOMO and LUMO distributions were changed by B and K co-doping, leading to improved separation of photoinduced carriers. The photocatalytic mechanism for water splitting over g-C3N4 semiconductor was speculated.
中文翻译:

B和K共掺杂g-C3N4的可回收熔盐合成用于光催化整体水蒸气分解
摘要 光催化全分解水是一种绿色高效的能源技术。由于光催化剂难以释放 O2,同时产生 H2 和 O2 是水分解的关键挑战。本研究采用可回收熔盐法合成了 B 和 K 共掺杂 g-C3N4 (B/Kg-C3N4) 催化剂。与 K 掺杂和原始 g-C3N4 样品相比,B/Kg-C3N4 在可见光照射下表现出最高的水蒸气分解光催化活性。H2和O2的产率达到约2:1,光照24小时后相应的产率分别为1.18和0.58μmol/h。B/Kg-C3N4 样品在 9 次循环后表现出良好的光催化稳定性。此外,从可重复回收的盐中获得的共掺杂 g-C3N4 催化剂,仍然表现出相当的光催化活性。基于使用 g-C3N4 构建的结构模型,B 和 K 共掺杂改变了 HOMO 和 LUMO 分布,从而改善了光生载流子的分离。推测了g-C3N4半导体上水分解的光催化机理。
更新日期:2021-01-01
中文翻译:

B和K共掺杂g-C3N4的可回收熔盐合成用于光催化整体水蒸气分解
摘要 光催化全分解水是一种绿色高效的能源技术。由于光催化剂难以释放 O2,同时产生 H2 和 O2 是水分解的关键挑战。本研究采用可回收熔盐法合成了 B 和 K 共掺杂 g-C3N4 (B/Kg-C3N4) 催化剂。与 K 掺杂和原始 g-C3N4 样品相比,B/Kg-C3N4 在可见光照射下表现出最高的水蒸气分解光催化活性。H2和O2的产率达到约2:1,光照24小时后相应的产率分别为1.18和0.58μmol/h。B/Kg-C3N4 样品在 9 次循环后表现出良好的光催化稳定性。此外,从可重复回收的盐中获得的共掺杂 g-C3N4 催化剂,仍然表现出相当的光催化活性。基于使用 g-C3N4 构建的结构模型,B 和 K 共掺杂改变了 HOMO 和 LUMO 分布,从而改善了光生载流子的分离。推测了g-C3N4半导体上水分解的光催化机理。











































 京公网安备 11010802027423号
京公网安备 11010802027423号