当前位置:
X-MOL 学术
›
J. Am. Chem. Soc.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Development of an Aryl Amination Catalyst with Broad Scope Guided by Consideration of Catalyst Stability
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2020-08-07 , DOI: 10.1021/jacs.0c06139
Scott D McCann 1 , Elaine C Reichert 1 , Pedro Luis Arrechea 1 , Stephen L Buchwald 1
Journal of the American Chemical Society ( IF 14.4 ) Pub Date : 2020-08-07 , DOI: 10.1021/jacs.0c06139
Scott D McCann 1 , Elaine C Reichert 1 , Pedro Luis Arrechea 1 , Stephen L Buchwald 1
Affiliation
|
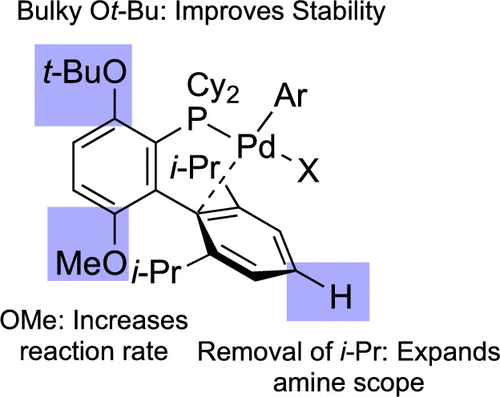
We have developed a new dialkylbiaryl monophosphine ligand, GPhos, that supports a palladium catalyst capable of promoting carbon-nitrogen cross-coupling reactions between a variety of primary amines and aryl halides; in many cases these reactions can be carried out at room temperature. The reaction development was guided by the idea that the productivity of catalysts employing BrettPhos-like ligands is limited by their lack of stability at room temperature. Specifically, it was hypothesized that primary amine and N-heteroaromatic substrates can displace the phosphine ligand, leading to the formation of catalytically dormant palladium complexes that reactivate only upon heating. This notion was supported by the synthesis and kinetic study of a putative off-cycle Pd complex. Consideration of this off-cycle species, together with the identification of substrate classes that are not effectively coupled at room temperature using previous catalysts, led to the design of a new dialkylbiaryl monophosphine ligand. An Ot-Bu substituent was added ortho to the dialkylphosphino group of the ligand framework to increase stability of the most active catalyst conformer. To offset the increased size of this substituent, we also removed the para i-Pr group of the non-phosphorous-containing ring, which allowed the catalyst to accommodate binding of even very large α-tertiary primary amine nucleophiles. Compared to previous catalysts, the GPhos-supported catalyst exhibits better reactivity both under ambient conditions and at elevated temperatures. Its use also allows for the coupling of a range of amine nucleophiles, including: (1) unhindered, (2) five-membered-ring N-heterocycle-containing, and (3) α-tertiary primary amines, each of which previously required a different catalyst to achieve optimal results.
中文翻译:

以催化剂稳定性为导向的广谱芳基胺化催化剂的开发
我们开发了一种新的二烷基联芳基单膦配体 GPhos,它支持钯催化剂,能够促进各种伯胺和芳基卤化物之间的碳氮交叉偶联反应;在许多情况下,这些反应可以在室温下进行。反应发展的指导思想是使用 BrettPhos 类配体的催化剂的生产率受到它们在室温下缺乏稳定性的限制。具体而言,假设伯胺和 N-杂芳族底物可以取代膦配体,导致形成催化休眠的钯配合物,该配合物仅在加热时重新活化。这一观点得到了假定的非循环 Pd 配合物的合成和动力学研究的支持。考虑到这种非循环物种,连同使用以前的催化剂在室温下不能有效偶联的底物类别的鉴定,导致了新的二烷基联芳基单膦配体的设计。Ot-Bu 取代基被添加到配体骨架的二烷基膦基的邻位,以增加最活跃的催化剂构象异构体的稳定性。为了抵消该取代基尺寸增加的影响,我们还去除了不含磷环的对位 i-Pr 基团,这使催化剂能够适应甚至非常大的 α-叔伯胺亲核试剂的结合。与以前的催化剂相比,GPhos 负载的催化剂在环境条件和高温下都表现出更好的反应性。它的使用还允许偶联一系列胺亲核试剂,包括:(1) 不受阻碍,
更新日期:2020-08-07
中文翻译:

以催化剂稳定性为导向的广谱芳基胺化催化剂的开发
我们开发了一种新的二烷基联芳基单膦配体 GPhos,它支持钯催化剂,能够促进各种伯胺和芳基卤化物之间的碳氮交叉偶联反应;在许多情况下,这些反应可以在室温下进行。反应发展的指导思想是使用 BrettPhos 类配体的催化剂的生产率受到它们在室温下缺乏稳定性的限制。具体而言,假设伯胺和 N-杂芳族底物可以取代膦配体,导致形成催化休眠的钯配合物,该配合物仅在加热时重新活化。这一观点得到了假定的非循环 Pd 配合物的合成和动力学研究的支持。考虑到这种非循环物种,连同使用以前的催化剂在室温下不能有效偶联的底物类别的鉴定,导致了新的二烷基联芳基单膦配体的设计。Ot-Bu 取代基被添加到配体骨架的二烷基膦基的邻位,以增加最活跃的催化剂构象异构体的稳定性。为了抵消该取代基尺寸增加的影响,我们还去除了不含磷环的对位 i-Pr 基团,这使催化剂能够适应甚至非常大的 α-叔伯胺亲核试剂的结合。与以前的催化剂相比,GPhos 负载的催化剂在环境条件和高温下都表现出更好的反应性。它的使用还允许偶联一系列胺亲核试剂,包括:(1) 不受阻碍,

































 京公网安备 11010802027423号
京公网安备 11010802027423号