Bioorganic & Medicinal Chemistry Letters ( IF 2.5 ) Pub Date : 2020-08-08 , DOI: 10.1016/j.bmcl.2020.127476 Kobirul Islam 1 , Kunal Pal 2 , Utsab Debnath 3 , R Sidick Basha 4 , Abu Taleb Khan 4 , Kuladip Jana 2 , Anup Kumar Misra 2
|
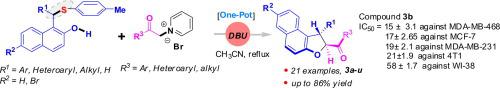
A series of 1,2-dihydronaphtho[2,1-b]furan derivatives were synthesized by cyclizing 1-(aryl/alkyl(arylthio)methyl)-naphthalen-2-ol and pyridinium bromides in the presence of 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) in very good yield. The synthesized compounds were evaluated for their anti-proliferative potential against human triple negative MDA-MB-468 and MCF-7 breast cancer cells and non-cancerous WI-38 cells (lung fibroblast cell) using MTT experiments. Among 21 synthesized compounds, three compounds (3a, 3b and 3 s) showed promising anti-cancer potential and compound 3b was found to have best anti-proliferative activities based on the results of several biochemical and microscopic experiments.
中文翻译:

(1,2-二氢萘并[2,1-b]呋喃-2-基)甲酮衍生物的抗癌潜力。
在1,8-二氮杂双环存在下,通过环化1-(芳基/烷基(芳硫基)甲基)-萘-2-醇和溴化吡啶鎓,合成了一系列1,2-二氢萘并[2,1- b ]呋喃衍生物[5.4.0]十一碳7烯(DBU)的收率非常好。使用MTT实验评估了合成的化合物对人三阴性MDA-MB-468和MCF-7乳腺癌细胞以及非癌性WI-38细胞(肺成纤维细胞)的抗增殖潜力。在21种合成化合物中,三种化合物(3a,3b和3 s)显示出有希望的抗癌潜力,并且根据一些生化和微观实验的结果,发现化合物3b具有最佳的抗增殖活性。































 京公网安备 11010802027423号
京公网安备 11010802027423号