当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Synthesis of 3-alkyl-6-methyl-1,2,4,5-tetrazines via a Sonogashira-type cross-coupling reaction.
Chemical Communications ( IF 4.3 ) Pub Date : 2020-08-05 , DOI: 10.1039/d0cc03482g Enric Ros 1 , Amparo Prades , Dominique Forson , Jacqueline Smyth , Xavier Verdaguer , Lluís Ribas de Pouplana , Antoni Riera
Chemical Communications ( IF 4.3 ) Pub Date : 2020-08-05 , DOI: 10.1039/d0cc03482g Enric Ros 1 , Amparo Prades , Dominique Forson , Jacqueline Smyth , Xavier Verdaguer , Lluís Ribas de Pouplana , Antoni Riera
Affiliation
|
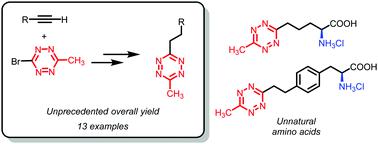
1,2,4,5-Tetrazines have become extremely useful tools in chemical biology. However, the synthesis of some challenging substrates such as asymmetrically disubstituted alkyltetrazines is still a limitation for the widespread use of this class of compounds. Herein we describe an efficient route to these compounds based on the Sonogashira coupling of 3-bromo-6-methyl-1,2,4,5-tetrazine and 3-bromo-6-phenyl-1,2,4,5-tetrazine with terminal alkynes. The preparation of the starting reagents has also been optimized. The alkynyl products have been used as intermediates for the synthesis of dialkyl-tetrazines through a sequence of hydrogenation and re-oxidation with unprecedented yields. The synthetic applicability of this new approach is showcased through the preparation of several unnatural amino acids bearing alkynyl- and alkyl-1,2,4,5-tetrazine fragments.
中文翻译:

通过Sonogashira型交叉偶联反应合成3-烷基-6-甲基-1,2,4,5-四嗪。
1,2,4,5-四嗪已成为化学生物学中极为有用的工具。然而,一些具有挑战性的底物如不对称双取代的烷基四嗪的合成仍然是这类化合物广泛使用的限制。本文中,我们基于3-溴-6-甲基-1,2,4,5-四嗪和3-溴-6-苯基-1,2,4,5-四嗪的Sonogashira偶联描述了制备这些化合物的有效途径与末端炔烃。起始试剂的制备也已优化。炔基产物已被用作中间体,通过一系列加氢和再氧化反应以前所未有的产率合成二烷基四嗪。通过制备几种带有炔基和烷基1,2,4,5-四嗪片段的非天然氨基酸,展示了这种新方法的合成适用性。
更新日期:2020-09-22
中文翻译:

通过Sonogashira型交叉偶联反应合成3-烷基-6-甲基-1,2,4,5-四嗪。
1,2,4,5-四嗪已成为化学生物学中极为有用的工具。然而,一些具有挑战性的底物如不对称双取代的烷基四嗪的合成仍然是这类化合物广泛使用的限制。本文中,我们基于3-溴-6-甲基-1,2,4,5-四嗪和3-溴-6-苯基-1,2,4,5-四嗪的Sonogashira偶联描述了制备这些化合物的有效途径与末端炔烃。起始试剂的制备也已优化。炔基产物已被用作中间体,通过一系列加氢和再氧化反应以前所未有的产率合成二烷基四嗪。通过制备几种带有炔基和烷基1,2,4,5-四嗪片段的非天然氨基酸,展示了这种新方法的合成适用性。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号