Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
β-Carboline Derivatives Tackling Malaria: Biological Evaluation and Docking Analysis.
ACS Omega ( IF 3.7 ) Pub Date : 2020-07-13 , DOI: 10.1021/acsomega.0c01256 Varun Gorki 1 , Neha Sylvia Walter 1 , Rahul Singh 2 , Monika Chauhan 3 , Neelima Dhingra 3 , Deepak B Salunke 2 , Sukhbir Kaur 1
ACS Omega ( IF 3.7 ) Pub Date : 2020-07-13 , DOI: 10.1021/acsomega.0c01256 Varun Gorki 1 , Neha Sylvia Walter 1 , Rahul Singh 2 , Monika Chauhan 3 , Neelima Dhingra 3 , Deepak B Salunke 2 , Sukhbir Kaur 1
Affiliation
|
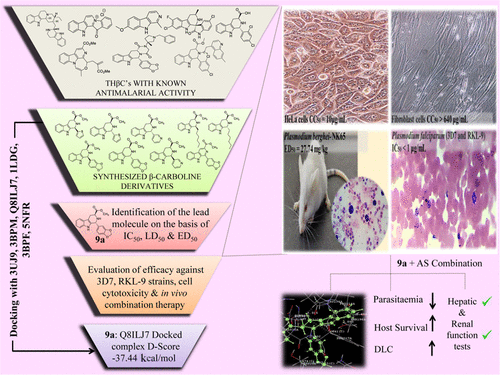
Increasing resistance to presently available antimalarial drugs urges the need to look for new promising compounds. The β-carboline moiety, present in several biologically active natural products and drugs, is an important scaffold for antimalarial drug discovery. The present study explores the antimalarial activity of a β-carboline derivative (1R,3S)-methyl 1-(benzo[d][1,3]dioxol-5-yl)-2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole-3-carboxylate (9a) alone in vitro against Plasmodium falciparum and in vivo in combination therapy with the standard drug artesunate against Plasmodium berghei. Compound 9a inhibited both 3D7 and RKL-9 strains of P. falciparum with half-maximal inhibitory concentration (IC50) < 1 μg/mL, respectively. The compound was nontoxic (50% cytotoxic concentration (CC50) > 640 μg/mL) to normal dermal fibroblasts. Selectivity index was >10 against both the strains. The compound exhibited considerable in vivo antimalarial activity (median effective dose (ED50) = 27.74 mg/kg) in monotherapy. The combination of 9a (100 mg/kg) and artesunate (50 mg/kg) resulted in 99.69% chemosuppression on day 5 along with a mean survival time of 25.8 ± 4.91 days with complete parasite clearance. Biochemical studies indicated the safety of the HIT compound to hepatic and renal functions of mice. Molecular docking also highlighted the suitability of 9a as a potential antimalarial candidate.
中文翻译:

β-咔啉衍生物对抗疟疾:生物学评价和对接分析。
对目前可用的抗疟药物的耐药性不断增加,迫切需要寻找新的有前途的化合物。 β-咔啉部分存在于多种生物活性天然产物和药物中,是抗疟药物发现的重要支架。本研究探讨了 β-咔啉衍生物 (1 R ,3 S )-methyl 1-(苯并[ d ][1,3]dioxol-5-yl)-2,3,4,9-tetrahydro 的抗疟活性-1 H-吡啶并[3,4- b ]吲哚-3-羧酸酯 ( 9a ) 单独在体外对抗恶性疟原虫,在体内与标准药物青蒿琥酯联合治疗对抗伯氏疟原虫。化合物9a分别抑制恶性疟原虫的3D7和RKL-9菌株,半数最大抑制浓度(IC 50 )<1μg/mL。该化合物对正常真皮成纤维细胞无毒(50%细胞毒性浓度(CC 50 )> 640 μg/mL)。针对这两种菌株的选择性指数均>10。该化合物在单一疗法中表现出相当大的体内抗疟活性(中位有效剂量(ED 50 )= 27.74 mg/kg)。 9a (100 mg/kg) 和青蒿琥酯 (50 mg/kg) 的组合在第 5 天产生 99.69% 的化疗抑制率,平均存活时间为 25.8 ± 4.91 天,寄生虫完全清除。生化研究表明 HIT 化合物对小鼠肝肾功能的安全性。分子对接还强调了9a作为潜在抗疟候选药物的适用性。
更新日期:2020-07-28
中文翻译:

β-咔啉衍生物对抗疟疾:生物学评价和对接分析。
对目前可用的抗疟药物的耐药性不断增加,迫切需要寻找新的有前途的化合物。 β-咔啉部分存在于多种生物活性天然产物和药物中,是抗疟药物发现的重要支架。本研究探讨了 β-咔啉衍生物 (1 R ,3 S )-methyl 1-(苯并[ d ][1,3]dioxol-5-yl)-2,3,4,9-tetrahydro 的抗疟活性-1 H-吡啶并[3,4- b ]吲哚-3-羧酸酯 ( 9a ) 单独在体外对抗恶性疟原虫,在体内与标准药物青蒿琥酯联合治疗对抗伯氏疟原虫。化合物9a分别抑制恶性疟原虫的3D7和RKL-9菌株,半数最大抑制浓度(IC 50 )<1μg/mL。该化合物对正常真皮成纤维细胞无毒(50%细胞毒性浓度(CC 50 )> 640 μg/mL)。针对这两种菌株的选择性指数均>10。该化合物在单一疗法中表现出相当大的体内抗疟活性(中位有效剂量(ED 50 )= 27.74 mg/kg)。 9a (100 mg/kg) 和青蒿琥酯 (50 mg/kg) 的组合在第 5 天产生 99.69% 的化疗抑制率,平均存活时间为 25.8 ± 4.91 天,寄生虫完全清除。生化研究表明 HIT 化合物对小鼠肝肾功能的安全性。分子对接还强调了9a作为潜在抗疟候选药物的适用性。


















































 京公网安备 11010802027423号
京公网安备 11010802027423号