当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Copper-Catalyzed Azide-Ynamide Cyclization to Generate α-Imino Copper Carbenes: Divergent and Enantioselective Access to Polycyclic N-Heterocycles.
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2020-07-04 , DOI: 10.1002/anie.202007206 Xin Liu,Ze-Shu Wang,Tong-Yi Zhai,Chen Luo,Yi-Ping Zhang,Yang-Bo Chen,Chao Deng,Rai-Shung Liu,Long-Wu Ye
Angewandte Chemie International Edition ( IF 16.1 ) Pub Date : 2020-07-04 , DOI: 10.1002/anie.202007206 Xin Liu,Ze-Shu Wang,Tong-Yi Zhai,Chen Luo,Yi-Ping Zhang,Yang-Bo Chen,Chao Deng,Rai-Shung Liu,Long-Wu Ye
|
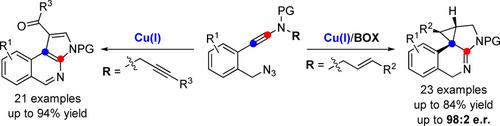
Here an efficient copper‐catalyzed cascade cyclization of azide‐ynamides via α‐imino copper carbene intermediates is reported, representing the first generation of α‐imino copper carbenes from alkynes. This protocol enables the practical and divergent synthesis of an array of polycyclic N‐heterocycles in generally good to excellent yields with broad substrate scope and excellent diastereoselectivities. Moreover, an asymmetric azide–ynamide cyclization has been achieved with high enantioselectivities (up to 98:2 e.r.) by employing BOX‐Cu complexes as chiral catalysts. Thus, this protocol constitutes the first example of an asymmetric azide–alkyne cyclization. The proposed mechanistic rationale for this cascade cyclization is further supported by theoretical calculations.
中文翻译:

铜催化的叠氮化物-亚酰胺环化生成α-氨基铜卡宾:对多环N-杂环的发散和对映选择性。
在此报道了通过α-亚氨基铜卡宾中间体对叠氮化物-酰胺类进行有效的铜催化级联环化反应,代表了炔烃的第一代α-亚氨基铜碳烯。该协议使多环N-杂环阵列的实用和发散性合成具有良好的收率,具有广泛的底物范围和出色的非对映选择性,通常具有良好的产率。此外,通过使用BOX-Cu络合物作为手性催化剂,已经实现了具有高对映选择性(高达98:2 er)的不对称叠氮化物-酰胺酰胺环化反应。因此,该协议构成了不对称叠氮化物-炔烃环化的第一个例子。理论计算进一步支持了该级联环化的机理原理。
更新日期:2020-07-04
中文翻译:

铜催化的叠氮化物-亚酰胺环化生成α-氨基铜卡宾:对多环N-杂环的发散和对映选择性。
在此报道了通过α-亚氨基铜卡宾中间体对叠氮化物-酰胺类进行有效的铜催化级联环化反应,代表了炔烃的第一代α-亚氨基铜碳烯。该协议使多环N-杂环阵列的实用和发散性合成具有良好的收率,具有广泛的底物范围和出色的非对映选择性,通常具有良好的产率。此外,通过使用BOX-Cu络合物作为手性催化剂,已经实现了具有高对映选择性(高达98:2 er)的不对称叠氮化物-酰胺酰胺环化反应。因此,该协议构成了不对称叠氮化物-炔烃环化的第一个例子。理论计算进一步支持了该级联环化的机理原理。













































 京公网安备 11010802027423号
京公网安备 11010802027423号