Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-05-05 , DOI: 10.1016/j.bmc.2020.115540
Mohsen Shekouhy 1 , Somaye Karimian 2 , Ali Moaddeli 3 , Zeinab Faghih 4 , Yousef Delshad 1 , Ali Khalafi-Nezhad 1
|
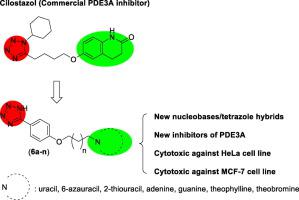
Spired by the chemical structure of Cilostazol, a selective phosphodiesterase 3A (PDE3A) inhibitor, several novel hybrid compounds of nucleobases (uracil, 6-azauracil, 2-thiuracil, adenine, guanine, theophylline and theobromine) and tetrazole were designed and successfully synthesized and their inhibitory effects on PDE3A as well as their cytotoxicity on HeLa and MCF-7 cancerous cell lines were studied. Obtained results show the linear correlation between the inhibitory effect of synthesized compounds and their cytotoxicity. In some cases, the PDE3A inhibitory effects of synthesized compounds are higher than the Cilostazol. Besides, compared to a standard anticancer drug methotrexate, some of the synthesized compounds showed the higher cytotoxicity against the HeLa and MCF-7 cancerous cell lines.
中文翻译:

核碱基/四唑杂化化合物的合成和生物学评估:一类新的3型磷酸二酯酶(PDE3)抑制剂。
受西洛他唑,选择性磷酸二酯酶3A(PDE3A)抑制剂的化学结构的刺激,设计并成功合成了几种新型核碱基(尿嘧啶,6-氮杂尿嘧啶,2-硫嘧啶,腺嘌呤,鸟嘌呤,茶碱和可可碱)和四唑的杂合化合物。研究了它们对PDE3A的抑制作用以及对HeLa和MCF-7癌细胞系的细胞毒性。所得结果表明合成化合物的抑制作用与其细胞毒性之间呈线性关系。在某些情况下,合成化合物对PDE3A的抑制作用高于西洛他唑。此外,与标准抗癌药甲氨蝶呤相比,一些合成的化合物对HeLa和MCF-7癌细胞具有更高的细胞毒性。

































 京公网安备 11010802027423号
京公网安备 11010802027423号