当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Asymmetric Synthesis of a Key Dextromethorphan Intermediate and Its Analogues Enabled by a New Cyclohexylamine Oxidase: Enzyme Discovery, Reaction Development, and Mechanistic Insight
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-03-23 , DOI: 10.1021/acs.joc.0c00469 Xiaofan Wu 1 , Zedu Huang 2, 3 , Zexu Wang 2, 3 , Zhining Li 2, 3 , Jiaqi Wang 2, 3 , Juan Lin 1, 4 , Fener Chen 2, 3
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2020-03-23 , DOI: 10.1021/acs.joc.0c00469 Xiaofan Wu 1 , Zedu Huang 2, 3 , Zexu Wang 2, 3 , Zhining Li 2, 3 , Jiaqi Wang 2, 3 , Juan Lin 1, 4 , Fener Chen 2, 3
Affiliation
|
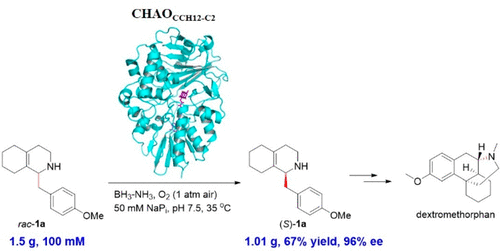
(S)-1-(4-Methoxybenzyl)-1,2,3,4,5,6,7,8-octahydroisoquinoline [(S)-1-(4-methoxybenzyl)-OHIQ, (S)-1a] is a key synthetic intermediate in the industrial production of dextromethorphan, one of the most widely used over-the-counter antitussives. We report here that a new cyclohexylamine oxidase discovered by genome mining, named CHAOCCH12-C2, was able to completely deracemize 100 mM 1a under Turner’s deracemization conditions to afford (S)-1a in 80% isolated yield and 99% ee at a semipreparative scale (0.4 mmol). When this biocatalytic reaction was scaled up to a gram scale (5.8 mmol), without reaction optimization (S)-1a was still isolated in 67% yield and 96% ee. The relatively higher kcat determined for CHAOCCH12-C2 was rationalized as one major factor rendering this enzyme capable of oxidizing 1a effectively at elevated substrate concentrations. Protein sequence alignment, analysis of our co-crystal structure of CHAOCCH12-C2 complexed with the product 1-(4-methoxybenzyl)-3,4,5,6,7,8-hexahydroisoquinoline [1-(4-methoxybenzyl)-HHIQ, 2a], and the structure-guided mutagenesis study together indicated L295 is one of the critical residues for this efficient enzymatic oxidation process and supported the presence of two cavities as well as a catalytically important “aromatic cage” formed by F342, Y433, and FAD. The synthetic applicability of CHAOCCH12-C2 was further underscored by the stereoselective synthesis of various enantioenriched 1-benzyl-OHIQ derivatives of potential pharmaceutical importance at a semipreparative scale.
中文翻译:

关键的右美沙芬中间体的不对称合成及其由新的环己基胺氧化酶实现的类似物:酶发现,反应发展和机制的见解。
(小号)-1-(4-甲氧基苄基)-1,2,3,4,5,6,7,8-全octahydroisoquinoline [(小号)-1-(4-甲氧基苄基)-OHIQ,(小号) - 1A ]是右美沙芬工业生产中的关键合成中间体,右美沙芬是最广泛使用的非处方镇咳药之一。我们在这里报告,通过基因组挖掘,名叫晁发现了一个新的环己胺氧化酶CCH12-C2,能够完全deracemize 100毫米1A特纳去外消旋条件下得到(小号) - 1A在80%的分离产率和99%EE在半制备规模(0.4毫摩尔)。当此生物催化反应放大至克级(5.8 mmol),而没有优化反应(S)-1a仍以67%的收率和96%的ee分离。确定CHAO CCH12-C2相对较高的k cat是使该酶能够在升高的底物浓度下有效氧化1a的主要因素之一。蛋白质序列比对,分析我们与产物1-(4-甲氧基苄基)-3,4,5,6,7,8-六氢异喹啉[1-(4-甲氧基苄基)-复合的CHAO CCH12-C2的共晶体结构HHIQ,2a],并且结构指导的诱变研究一起表明L295是该有效酶促氧化过程的关键残基之一,并支持存在两个腔以及F342,Y433和FAD形成的催化重要的“芳族笼”。CHAO CCH12-C2的合成适用性在半制备规模上通过立体选择性合成了各种具有潜在药学重要性的对映体富集的1-苄基-OHIQ衍生物而得到进一步强调。
更新日期:2020-04-24
中文翻译:

关键的右美沙芬中间体的不对称合成及其由新的环己基胺氧化酶实现的类似物:酶发现,反应发展和机制的见解。
(小号)-1-(4-甲氧基苄基)-1,2,3,4,5,6,7,8-全octahydroisoquinoline [(小号)-1-(4-甲氧基苄基)-OHIQ,(小号) - 1A ]是右美沙芬工业生产中的关键合成中间体,右美沙芬是最广泛使用的非处方镇咳药之一。我们在这里报告,通过基因组挖掘,名叫晁发现了一个新的环己胺氧化酶CCH12-C2,能够完全deracemize 100毫米1A特纳去外消旋条件下得到(小号) - 1A在80%的分离产率和99%EE在半制备规模(0.4毫摩尔)。当此生物催化反应放大至克级(5.8 mmol),而没有优化反应(S)-1a仍以67%的收率和96%的ee分离。确定CHAO CCH12-C2相对较高的k cat是使该酶能够在升高的底物浓度下有效氧化1a的主要因素之一。蛋白质序列比对,分析我们与产物1-(4-甲氧基苄基)-3,4,5,6,7,8-六氢异喹啉[1-(4-甲氧基苄基)-复合的CHAO CCH12-C2的共晶体结构HHIQ,2a],并且结构指导的诱变研究一起表明L295是该有效酶促氧化过程的关键残基之一,并支持存在两个腔以及F342,Y433和FAD形成的催化重要的“芳族笼”。CHAO CCH12-C2的合成适用性在半制备规模上通过立体选择性合成了各种具有潜在药学重要性的对映体富集的1-苄基-OHIQ衍生物而得到进一步强调。































 京公网安备 11010802027423号
京公网安备 11010802027423号