当前位置:
X-MOL 学术
›
Bioorg. Med. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
An increase in side-group hydrophobicity largely improves the potency of ritonavir-like inhibitors of CYP3A4.
Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-01-31 , DOI: 10.1016/j.bmc.2020.115349
Eric R Samuels 1 , Irina F Sevrioukova 2
Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2020-01-31 , DOI: 10.1016/j.bmc.2020.115349
Eric R Samuels 1 , Irina F Sevrioukova 2
Affiliation
|
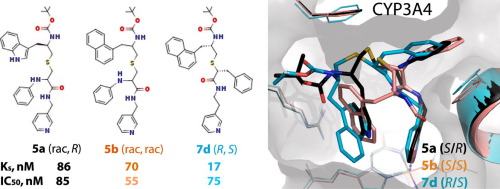
Identification of structural determinants required for potent inhibition of drug-metabolizing cytochrome P450 3A4 (CYP3A4) could help develop safer drugs and more effective pharmacoenhancers. We utilize a rational inhibitor design to decipher structure-activity relationships in analogues of ritonavir, a highly potent CYP3A4 inhibitor marketed as pharmacoenhancer. Analysis of compounds with the R1 side-group as phenyl or naphthalene and R2 as indole or naphthalene in different stereo configuration showed that (i) analogues with the R2-naphthalene tend to bind tighter and inhibit CYP3A4 more potently than the R2-phenyl/indole containing counterparts; (ii) stereochemistry becomes a more important contributing factor, as the bulky side-groups limit the ability to optimize protein-ligand interactions; (iii) the relationship between the R1/R2 configuration and preferential binding to CYP3A4 is complex and depends on the side-group functionality/interplay and backbone spacing; and (iv) three inhibitors, 5a-b and 7d, were superior to ritonavir (IC50 of 0.055-0.085 μM vs. 0.130 μM, respectively).
中文翻译:

侧基疏水性的增加大大改善了CYP3A4的利托那韦样抑制剂的效价。
确定有效抑制药物代谢性细胞色素P450 3A4(CYP3A4)所需的结构决定簇可以帮助开发更安全的药物和更有效的药物增强剂。我们利用合理的抑制剂设计来解释利托那韦类似物的结构活性关系,利托那韦是一种作为药物增强剂销售的高效CYP3A4抑制剂。分析具有不同立体结构的R1侧基为苯基或萘,R2为吲哚或萘的化合物显示:(i)与R2-萘类似的化合物比R2-苯基/吲哚具有更紧密的结合力和更强的CYP3A4抑制力包含同行 (ii)立体化学成为一个更重要的贡献因素,因为庞大的侧基限制了优化蛋白质-配体相互作用的能力;(iii)R1 / R2构型与优先结合CYP3A4的关系很复杂,并且取决于侧基的功能性/相互作用和主链间距;(iv)三种抑制剂5a-b和7d优于利托那韦(IC50分别为0.055-0.085μM和0.130μM)。
更新日期:2020-01-31
中文翻译:

侧基疏水性的增加大大改善了CYP3A4的利托那韦样抑制剂的效价。
确定有效抑制药物代谢性细胞色素P450 3A4(CYP3A4)所需的结构决定簇可以帮助开发更安全的药物和更有效的药物增强剂。我们利用合理的抑制剂设计来解释利托那韦类似物的结构活性关系,利托那韦是一种作为药物增强剂销售的高效CYP3A4抑制剂。分析具有不同立体结构的R1侧基为苯基或萘,R2为吲哚或萘的化合物显示:(i)与R2-萘类似的化合物比R2-苯基/吲哚具有更紧密的结合力和更强的CYP3A4抑制力包含同行 (ii)立体化学成为一个更重要的贡献因素,因为庞大的侧基限制了优化蛋白质-配体相互作用的能力;(iii)R1 / R2构型与优先结合CYP3A4的关系很复杂,并且取决于侧基的功能性/相互作用和主链间距;(iv)三种抑制剂5a-b和7d优于利托那韦(IC50分别为0.055-0.085μM和0.130μM)。

































 京公网安备 11010802027423号
京公网安备 11010802027423号