当前位置:
X-MOL 学术
›
J. Heterocycl. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
1,3‐Oxazole‐isoniazid hybrids: Synthesis, antitubercular activity, and their docking studies
Journal of Heterocyclic Chemistry ( IF 2.0 ) Pub Date : 2020-01-17 , DOI: 10.1002/jhet.3893
Shailesh R. Shah 1 , Kanubhai D. Katariya 1
Journal of Heterocyclic Chemistry ( IF 2.0 ) Pub Date : 2020-01-17 , DOI: 10.1002/jhet.3893
Shailesh R. Shah 1 , Kanubhai D. Katariya 1
Affiliation
|
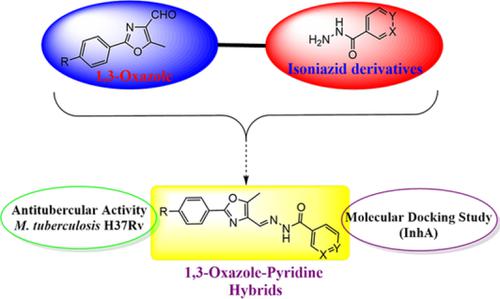
A series of novel N′‐([2‐aryl‐5‐methyl‐1,3‐oxazole‐4‐yl]methylene)isonicotino/nicotino hydrazides 10a‐l were prepared by the condensation reaction of 2‐aryl‐5‐methyl‐1,3‐oxazole‐4‐carbaldehydes 8a‐f with the corresponding isonicotino/nicotino hydrazides 9a/9b. The structures of the new compounds were elucidated by various spectroanalytical techniques, including IR, 1H NMR, 13C NMR, elemental (C,H,N), and mass analysis. All the newly prepared INH‐1,3‐oxazole hybrids were evaluated for their in vitro antitubercular activity against Mycobacterium tuberculosis H37Rv. Among all the synthesized hybrids, compounds 10c and 10i derivatives displayed highest antitubercular activity with minimal inhibitory concentration 1.56 μg/mL. Further, molecular docking studies against the InhA enzyme were carried out to understand the interactions between potent hybrids and the target enzyme. Thus, these kind hybrids have the potentiality for the discovery of new antitubercular agents for deployment in the control and eradication of tuberculosis.
中文翻译:

1,3-恶唑异烟肼杂化物:合成,抗结核活性及其对接研究
通过2-芳基-5-甲基的缩合反应制备了一系列新型的N '-([[2-芳基-5-甲基-1,3-恶唑-4-基]亚甲基]异烟肼/烟碱酰肼10a-1。 -1,3-恶唑-4-甲醛8a-f和相应的异烟肼/烟碱酰肼9a / 9b。通过各种光谱分析技术阐明了新化合物的结构,包括IR,1 H NMR,13 C NMR,元素(C,H,N)和质量分析。所有新制备的INH基-1,3-唑杂交种的评价在 体外对抗结核活性结核分枝杆菌分枝杆菌。在所有合成的杂种中,化合物10c和10i衍生物显示出最高的抗结核活性,最小抑菌浓度为1.56μg/ mL。此外,进行了针对InhA酶的分子对接研究,以了解有效杂种与目标酶之间的相互作用。因此,这些杂种具有发现新的抗结核药物用于控制和根除结核病的潜力。
更新日期:2020-01-17
中文翻译:

1,3-恶唑异烟肼杂化物:合成,抗结核活性及其对接研究
通过2-芳基-5-甲基的缩合反应制备了一系列新型的N '-([[2-芳基-5-甲基-1,3-恶唑-4-基]亚甲基]异烟肼/烟碱酰肼10a-1。 -1,3-恶唑-4-甲醛8a-f和相应的异烟肼/烟碱酰肼9a / 9b。通过各种光谱分析技术阐明了新化合物的结构,包括IR,1 H NMR,13 C NMR,元素(C,H,N)和质量分析。所有新制备的INH基-1,3-唑杂交种的评价在 体外对抗结核活性结核分枝杆菌分枝杆菌。在所有合成的杂种中,化合物10c和10i衍生物显示出最高的抗结核活性,最小抑菌浓度为1.56μg/ mL。此外,进行了针对InhA酶的分子对接研究,以了解有效杂种与目标酶之间的相互作用。因此,这些杂种具有发现新的抗结核药物用于控制和根除结核病的潜力。

































 京公网安备 11010802027423号
京公网安备 11010802027423号