Abstract
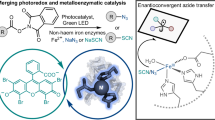
Enzymes are increasingly explored for use in asymmetric synthesis1,2,3, but their applications are generally limited by the reactions available to naturally occurring enzymes. Recently, interest in photocatalysis4 has spurred the discovery of novel reactivity from known enzymes5. However, so far photoinduced enzymatic catalysis6 has not been used for the cross-coupling of two molecules. For example, the intermolecular coupling of alkenes with α-halo carbonyl compounds through a visible-light-induced radical hydroalkylation, which could provide access to important γ-chiral carbonyl compounds, has not yet been achieved by enzymes. The major challenges are the inherent poor photoreactivity of enzymes and the difficulty in achieving stereochemical control of the remote prochiral radical intermediate7. Here we report a visible-light-induced intermolecular radical hydroalkylation of terminal alkenes that does not occur naturally, catalysed by an ‘ene’ reductase using readily available α-halo carbonyl compounds as reactants. This method provides an efficient approach to the synthesis of various carbonyl compounds bearing a γ-stereocentre with excellent yields and enantioselectivities (up to 99 per cent yield with 99 per cent enantiomeric excess), which otherwise are difficult to access using chemocatalysis. Mechanistic studies suggest that the formation of the complex of the substrates (α-halo carbonyl compounds) and the ‘ene’ reductase triggers the enantioselective photoinduced radical reaction. Our work further expands the reactivity repertoire of biocatalytic, synthetically useful asymmetric transformations by the merger of photocatalysis and enzyme catalysis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. The X-ray crystallographic coordinate for the structure of 3f reported in this article has been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition number CCDC 1989831.
References
Chen, K. & Arnold, F. H. Engineering new catalytic activities in enzymes. Nat. Catal. 3, 203–213 (2020).
Reetz, M. T. Laboratory evolution of stereoselective enzymes: a prolific source of catalysts for asymmetric reactions. Angew. Chem. Int. Ed. 50, 138–174 (2011).
Devine, P. N. et al. Extending the application of biocatalysis to meet the challenges of drug development. Nat. Rev. Chem. 2, 409–421 (2018).
Stephenson, C. R. J., Yoon, T. P. & MacMillan, D. W. C. (eds) Visible Light Photocatalysis in Organic Chemistry (Wiley-VCH, 2018).
Sandoval, B. A. & Hyster, T. K. Emerging strategies for expanding the toolbox of enzymes in biocatalysis. Curr. Opin. Chem. Biol. 55, 45–51 (2020).
Schmermund, L. et al. Photo-biocatalysis: biotransformations in the presence of light. ACS Catal. 9, 4115–4144 (2019).
Zimmerman, J. & Sibi, M. P. Enantioselective radical reactions. Top. Curr. Chem. 263, 107–162 (2006).
Lo, J. C., Gui, J., Yabe, Y., Pan, C.-M. & Baran, P. S. Functionalized olefin cross-coupling to construct carbon–carbon bonds. Nature 516, 343–348 (2014).
Studer, A. & Curran, D. P. Catalysis of radical reactions: a radical chemistry perspective. Angew. Chem. Int. Ed. 55, 58–102 (2016).
Huo, H., Harms, K. & Meggers, E. Catalytic, enantioselective addition of alkyl radicals to alkenes via visible-light-activated photoredox catalysis with a chiral rhodium complex. J. Am. Chem. Soc. 138, 6936–6939 (2016).
Capacci, A. G., Malinowski, J. T., McAlpine, N. J., Kuhne, J. & MacMillan, D. W. C. Direct, enantioselective α-alkylation of aldehydes using simple olefins. Nat. Chem. 9, 1073–1077 (2017).
Yegdaneh, A., Putchakarn, S., Yuenyongsawad, S., Ghannadi, A. & Plubrukarn, A. 3-Oxoabolene and 1-oxocurcuphenol, aromatic bisabolanes from the sponge Myrmekioderma sp. Nat. Prod. Commun. 8, 1355–1357 (2013).
Li, X.-D., Li, X.-M., Xu, G.-M., Zhang, P. & Wang, B.-G. Antimicrobial phenolic bisabolanes and related derivatives from Penicillium aculeatum SD-321, a deep sea sediment-derived fungus. J. Nat. Prod. 78, 844–849 (2015).
Liao, G. et al. The development of piperidinones as potent MDM2-P53 protein–protein interaction inhibitors for cancer therapy. Eur. J. Med. Chem. 159, 1–9 (2018).
Das, B., Shinde, D. B., Kanth, B. S., Kamle, A. & Kumar, C. G. Total synthesis of racemic and (R) and (S)-4-methoxyalkanoic acids and their antifungal activity. Eur. J. Med. Chem. 46, 3124–3129 (2011).
Alfaro Blasco, M. & Gröger, H. Enzymatic resolution of racemates with a ‘remote’ stereogenic center as an efficient tool in drug, flavor and vitamin synthesis. Bioorg. Med. Chem. 22, 5539–5546 (2014).
Gagnon, C. et al. Biocatalytic synthesis of planar chiral macrocycles. Science 367, 917–921 (2020).
Huffman, M. A. et al. Design of an in vitro biocatalytic cascade for the manufacture of islatravir. Science 366, 1255–1259 (2019).
Toogood, H. S. & Scrutton, N. S. Discovery, characterisation, engineering and applications of ene-reductases for industrial biocatalysis. ACS Catal. 8, 3532–3549 (2018).
Zultanski, S. L. & Fu, G. C. Catalytic asymmetric γ-alkylation of carbonyl compounds via stereoconvergent Suzuki cross-couplings. J. Am. Chem. Soc. 133, 15362–15364 (2011).
Emmanuel, M. A., Greenberg, N. R., Oblinsky, D. G. & Hyster, T. K. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light. Nature 540, 414–417 (2016).
Biegasiewicz, K. F., Cooper, S. J., Emmanuel, M. A., Miller, D. C. & Hyster, T. K. Catalytic promiscuity enabled by photoredox catalysis in nicotinamide-dependent oxidoreductases. Nat. Chem. 10, 770–775 (2018).
Litman, Z. C., Wang, Y., Zhao, H. & Hartwig, J. F. Cooperative asymmetric reactions combining photocatalysis and enzymatic catalysis. Nature 560, 355–359 (2018).
Biegasiewicz, K. F. et al. Photoexcitation of flavoenzymes enables a stereoselective radical cyclization. Science 364, 1166–1169 (2019).
Black, M. J. et al. Asymmetric redox-neutral radical cyclization catalysed by flavin-dependent ‘ene’-reductases. Nat. Chem. 12, 71–75 (2020).
Sorigué, D. et al. An algal photoenzyme converts fatty acids to hydrocarbons. Science 357, 903–907 (2017).
Xu, J. et al. Light-driven kinetic resolution of α-functionalized carboxylic acids enabled by an engineered fatty acid photodecarboxylase. Angew. Chem. Int. Ed. 58, 8474–8478 (2019).
Brimioulle, R., Lenhart, D., Maturi, M. M. & Bach, T. Enantioselective catalysis of photochemical reactions. Angew. Chem. Int. Ed. 54, 3872–3890 (2015).
Rudroff, F. et al. Opportunities and challenges for combining chemo- and biocatalysis. Nat. Catal. 1, 12–22 (2018).
Sandoval, B. A., Meichan, A. J. & Hyster, T. K. Enantioselective hydrogen atom transfer: discovery of catalytic promiscuity in flavin-dependent ‘ene’-reductases. J. Am. Chem. Soc. 139, 11313–11316 (2017).
Doyle, A. G. & Jacobsen, E. N. Small-molecule H-bond donors in asymmetric catalysis. Chem. Rev. 107, 5713–5743 (2007).
Crisenza, G. E. M., Mazzarella, D. & Melchiorre, P. Synthetic methods driven by the photoactivity of electron donor–acceptor complexes. J. Am. Chem. Soc. 142, 5461–5476 (2020).
Beckwith, A. L. J. & Bowry, V. W. Kinetics of reactions of cyclopropylcarbinyl radicals and alkoxycarbonyl radicals containing stabilizing substituents: implications for their use as radical clocks. J. Am. Chem. Soc. 116, 2710–2716 (1994).
Acknowledgements
This work was supported by the US Department of Energy (DE-SC0018420). NMR data was collected in the Carl R. Woese Institute for Genomic Biology Core on a 600-MHz NMR funded by NIH grant number S10-RR028833. We thank T. Woods for assistance with X-ray diffraction studies and X. Guan for help with NMR analysis. X.H. acknowledges partial support from the Shen Postdoctoral Fellowship.
Author information
Authors and Affiliations
Contributions
H.Z. coordinated the project. X.H. and H.Z. conceived the project and designed the experiments. X.H., Y.W. and G.J. performed the experiments. B.W. and J.F. carried out the computational studies. X.H, B.W. and H.Z. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Frank Hollman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file contains Supplementary Figs. 1-19, Supplementary Tables 1-7, Supplementary Methods and Supplementary References.
Rights and permissions
About this article
Cite this article
Huang, X., Wang, B., Wang, Y. et al. Photoenzymatic enantioselective intermolecular radical hydroalkylation. Nature 584, 69–74 (2020). https://doi.org/10.1038/s41586-020-2406-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2406-6